The FDA will explore allowing drug importation to counteract egregious generic price hikes.
Most of President Trump’s much-hyped agenda to rein in drug prices has been underwhelming so far, but the Food and Drug Administration announced on Thursday it will explore one potentially significant idea: drug importation.
The FDA said that it was creating a working group to look into the idea, which has been floated for a while, to address certain generic drug price hikes. The gist of it seems to be that if a drug company increases the price for a generic drug for which there are no real substitutes, the agency could allow drugs from other countries to be imported. With some added competition, prices would hopefully come down.
Right now, in the above circumstances and when there isn’t an actual drug shortage, “we don’t have a good policy solution,” Scott Gottlieb, the FDA commissioner, told me Thursday in a brief phone interview.
The policy would be applied in limited circumstances — only for generic drugs and only temporarily, it appears — and could therefore have only a limited effect, but experts still say it’s a good idea.
“I think this is a smart move for the administration,” Rachel Sachs, a law professor at Washington University in St. Louis, who has generally been unimpressed by what the Trump administration has proposed on drug prices, told me.
It’s just a working group for now. But if the FDA follows through, it would be one of the few policies pursued by the Trump administration that the pharmaceutical industry doesn’t care for. The brand-name drug lobby has warned of the “dangers of drug importation” in the past — although the proposal laid out by the FDA would apply to generic drugs only, it seems.
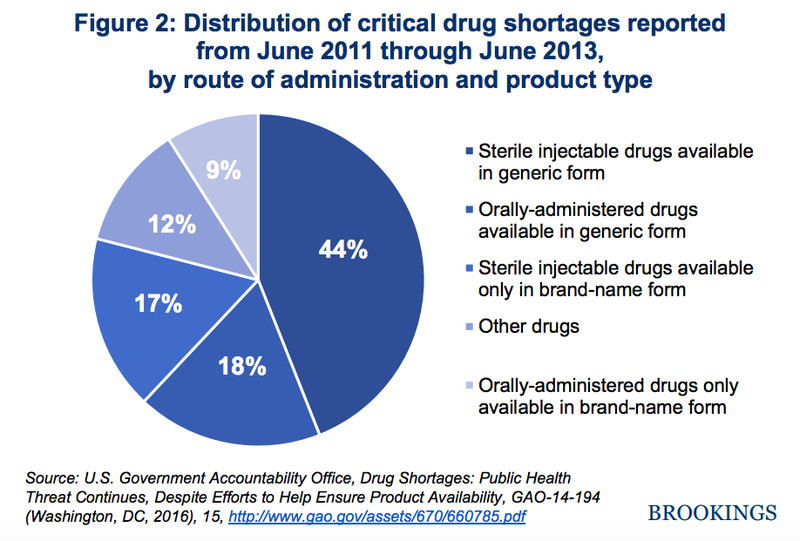
Generic drugs without competitors are a real problem. Drug shortages have been on the rise in the United States — tripling from 2007 to 2012, according to the Brookings Institution — and generics are responsible for many of them.
 Brookings Institution
Brookings InstitutionEgregious generic price hikes signal a breakdown in the prescription drug market. Generics are supposed to introduce competition, by entering the market with brand-name drugs that have the same therapeutic use, but instead, we have seen several notable examples of when a generic drug’s price was significantly raised and no competitor existed to counter it.
That is what Martin Shkreli’s drug company did before he went to prison, as did Valeant Pharmaceuticals, the executives of which were dragged before Congress a couple years ago.
The Brooking Institution examined in May 2017 whether allowing drug importation could address high generic drug prices, so the FDA’s interest didn’t come out of nowhere. Proposals like this have been around for a while.
“The idea of expanding international competition to address price spikes in generic drugs is an excellent one,” the authors wrote, though they wanted to go further than the Trump administration seems willing to. The FDA’s announcement signals that importation would be temporary, for example, while the Brookings experts discourage temporary measures.
Still, it’s an encouraging development, given how otherwise disappointing the Trump administration’s drug pricing agenda has been. Gottlieb has stood out to many health care experts for his willingness to pursue interesting policy ideas. President Trump has indicated importation as a policy he would support, though not very regularly.
Gottlieb told me that a key part of this, and the reason he convened the working group, is defining what situations are egregious enough that they warrant allowing importation.
“I think it’s gonna be challenging to define that,” he said. “We need reproducible definitions with clear boundaries.”
The hope is to have more substantive policy specifics by the end of the year. Drug importation could potentially be done through administrative action, Gottlieb said, though he allowed that it could require Congress to pass legislation.
Read More
https://cdn.vox-cdn.com/community_logos/52517/voxv.png